AAA-SHAPE Randomized Controlled Trial
Investigational device, limited by Federal (or United States) law to investigational use.
The aorta, your largest artery, is the main pathway which carries blood from your heart to the rest of your body. Various disorders and conditions that affect the aorta are classified as aortic diseases.
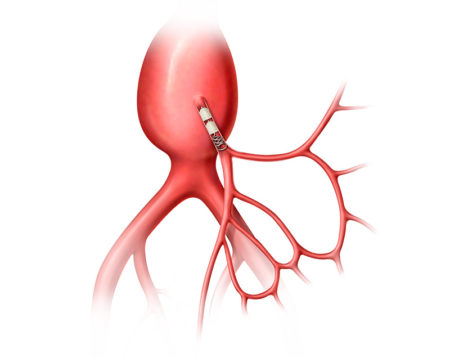
Aortic Aneurysms are a weakened area in the wall of the aorta, where enlargement or swelling is observed. If left untreated, the weakened aortic wall can balloon and potentially rupture.
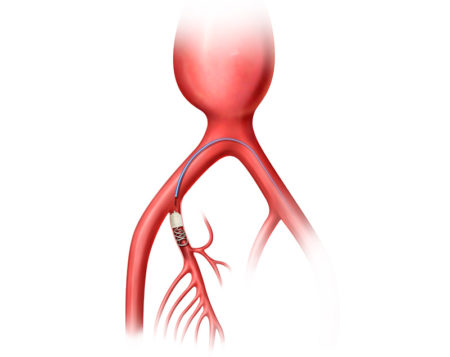
Aortic aneurysms can be treated through open surgical repair, or through an endovascular (less invasive), life-saving procedure called EVAR (Endovascular Aneurysm Repair).
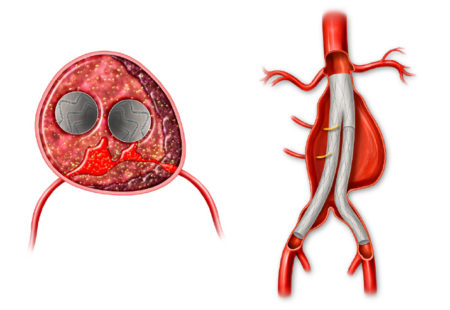
However, not all aneurysms respond to EVAR, failing to shrink in spite of treatment. This unmet clinical need is significant and can be associated with higher risks of complications, rehospitalization, increased costs, and increased mortality.
Shape Memory Medical is dedicated to patients and physicians by developing new aortic restorative therapies using our novel smart polymer technology through various clinical studies.
AAA-SHAPE Pivotal Trial
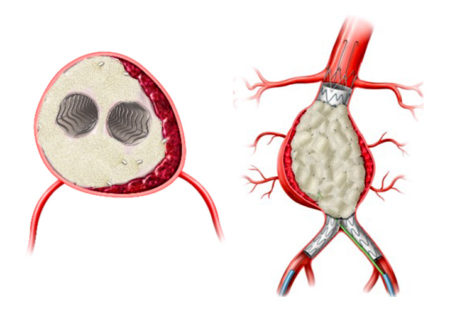
The AAA-SHAPE Pivotal Trial is a prospective, multicenter, randomized, open-label trial to determine safety and effectiveness of the IMPEDE-FX RapidFill Device to improve abdominal aortic aneurysm (AAA) sac behavior with used with elective endovascular aneurysm repair (EVAR).
The trial will enroll 180 patients with infrarenal AAA across 50 sites in the U.S., Europe, and New Zealand. Study participants will be randomized 2:1; either to EVAR plus sac management with the IMPEDE-FX RapidFill (the treatment arm) or to standard EVAR (the control arm). Key endpoints will compare sac diameter and volume change, endoleak rates, secondary interventions, and mortality through five years.

Abdominal Aortic Aneurysm Sac Healing and Prevention of Expansion (AAA-SHAPE)
The AAA-SHAPE Pivotal Trial is intended to determine the safety and effectiveness of IMPEDE-FX RapidFill to increase the percentage of subjects with shrinkage of the abdominal aortic aneurysm sac when used as an adjunct to on-label endovascular aneurysm repair (EVAR) stent graft treatment in trial subjects considered candidates for elective EVAR.