AAA-SHAPE Early Feasibility Studies
Investigational device, limited by Federal (or United States) law to investigational use.
The aorta, your largest artery, is the main pathway which carries blood from your heart to the rest of your body. Various disorders and conditions that affect the aorta are classified as aortic diseases.
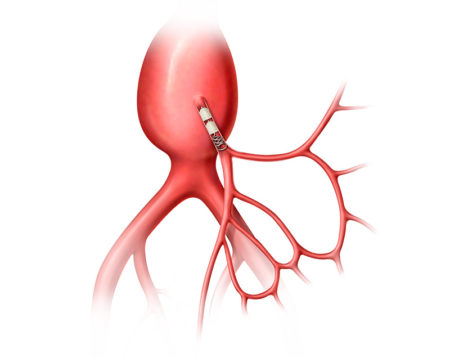
Aortic Aneurysms are a weakened area in the wall of the aorta, where enlargement or swelling is observed. If left untreated, the weakened aortic wall can balloon and potentially rupture.
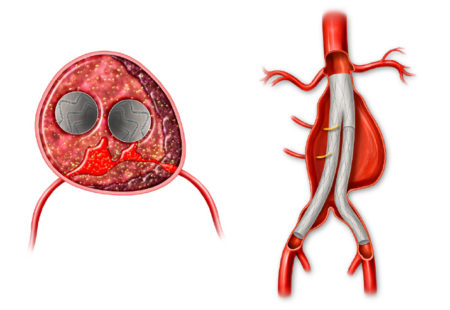
Aortic aneurysms can be treated through open surgical repair, or through an endovascular (less invasive), life-saving procedure called EVAR (Endovascular Aneurysm Repair).
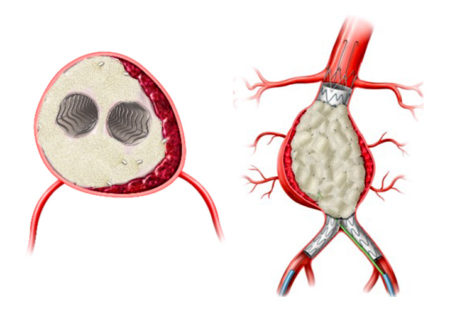
However, not all aneurysms respond to EVAR, failing to shrink in spite of treatment. This unmet clinical need is significant and can be associated with higher risks of complications, rehospitalization, increased costs, and increased mortality.
Shape Memory Medical is dedicated to patients and physicians by developing new aortic restorative therapies using our novel smart polymer technology through various clinical studies.
AAA-SHAPE Safety & Early Feasibility Studies
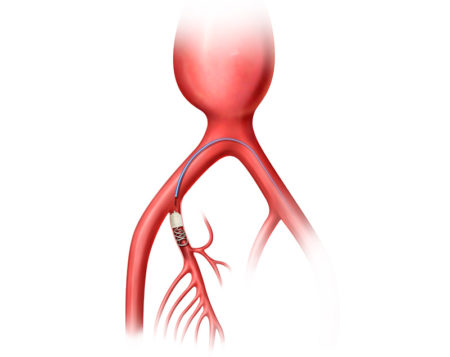
AAA-SHAPE FIH is the first prospective clinical trial to evaluate IMPEDE-FX RapidFill in AAA patients and its potential to remodel the AAA sac and improve aneurysm sac regression following EVAR. The study has enrolled 35 patients across three centers in the Netherlands, and two centers in New Zealand.
One year follow-up was completed in 2023.

Abdominal Aortic Aneurysm Sac Healing and Prevention of Endoleaks (AAA-SHAPE)
AAA-SHAPE NZ and NLD are prospective, multicenter safety and early feasibility trials to determine the safety and efficacy of the IMPEDE-FX Embolization Plug and/or IMPEDE-FX RapidFill to fill an abdominal aortic aneurysm (AAA) sac outside of an endovascular aneurysm repair (EVAR) stent graft.
Clinical Trial Presentations

Excitement builds over the AAA-SHAPE Pivotal Trial following 2-year early feasibility data with the novel shape memory polymer
Andrew Holden, MBChB and Dai Yamanouchi, MD, PhD – Charing Cross Symposium 2025
At this year’s Charing Cross (CX) Symposium (23–25 April, London, UK), two leading figures in the endovascular aneurysm repair (EVAR) field—Andrew Holden (Auckland City Hospital, Auckland, New Zealand) and Dai Yamanouchi (University of Wisconsin, Madison, USA)—sat down to discuss the potential benefits of active sac management with the investigational IMPEDE-FX RapidFill device, remarking that sac shrinkage is key for a long term, durable EVAR procedure.

CX co-chairs host discussion on studies looking at pre-emptive AAA sac management with the novel shape memory polymer and its potential to improve post-EVAR sac regression.
Dittmar Bӧckler, MD, Andrew Holden, MBChB, and Marc Schermerhorn, MD – Charing Cross Symposium 2024
Professor Holden highlighted data from the AAA-SHAPE early feasibility studies that evaluated the IMPEDE-FX Embolization Plug for management of AAA sacs at the time of EVAR. This novel technology is comprised of shape memory polymer, a porous polyurethane scaffold that supports rapid conversion to organized thrombus, that Professor Holden reports could lead to sac shrinkage. Schermerhorn also highlighted the need for evidence from a randomized clinical trial and announced the initiation of the AAA-SHAPE Pivotal Trial.

Active sac management represents a “step forward” for EVAR
Jan Heyligers, MD, PhD, & Michel Reijnen, MD, PhD – VEITH Symposium 2023
“Despite significant technological improvements since its inception, endovascular repair of abdominal aortic aneurysms (AAA) is still associated with some of the same problems we experienced nearly 30 years ago,” Michel Reijnen (Arnhem, The Netherlands) remarked at the VEITHsymposium 2023 (14–18 November, New York, USA). One of these issues—and a significant unmet need in the aortic field—is the failure of an AAA to regress post-endovascular aneurysm repair (EVAR).

Shape Memory Polymer addresses an “unmet need” for EVAR
Andrew Holden, MBChB, FRANZCR, EBIR & Michel Reijnen, MD, PhD – Charing Cross Symposium 2022
“When you compare it to other embolic agents it is completely different.”